Sommaire
Introduction
Through its proposal for a regulation on certain plants obtained by new genomic techniques (NGTs), the European Commission seeks to fundamentally reshape the regulatory framework applicable to plant innovation. The proposal is based on a differentiated approach: certain plants, by reason of the nature of the genetic modifications introduced, would be excluded from the traditional scope of genetically modified organisms (GMOs), while others would remain subject to a specific regulatory regime.
This shift in regulatory boundaries is far from merely technical. It directly affects the legal qualification of products, the predictability of applicable rules, and the operational flexibility available to economic operators within the European market. The reform thus raises a central question: according to which criteria should EU law henceforth distinguish between non-regulated innovation and regulated innovation?
What are “new genomic techniques”?
New genomic techniques (NGTs) refer to recent methods enabling targeted modification of an organism’s genome. In other words, they allow precise intervention on a specific segment of DNA. The most well-known tool is currently CRISPR-Cas9, which operates through an enzyme that cuts DNA and a guide system directing that enzyme to the exact location where the modification is to be made.
These techniques may produce highly diverse outcomes. They may, for instance, introduce minor DNA alterations, deactivate a gene, modify the protein encoded by that gene, or introduce a new genetic trait. NGTs therefore do not constitute a single technique, but rather a set of tools capable of producing varied effects, which complicates their legal qualification.
The current legal status of NGTs
Following an action brought in 2015 by several associations, the Court of Justice of the European Union (CJEU) was called upon to clarify the legal status of techniques now referred to as NGTs (CJEU, 25 July 2018, Case C-528/16).
In its decision of 25 July 2018, the Court held that organisms obtained through new mutagenesis techniques fall, in principle, within the definition of GMOs under Directive 2001/18/EC. It further recalled that only certain conventional mutagenesis techniques are exempt, as they have long been used and are considered to have an established safety record. NGTs, as more recent techniques, do not benefit from this exemption and therefore remain subject to the GMO regulatory framework under current law.
In practice, this assimilation has generated legal uncertainty, insofar as plants with identical characteristics may fall under different regulatory regimes depending on the technique used. Operators are thus exposed to the risk of reclassification as GMOs, with direct implications for regulatory obligations, time-to-market, and the security of their investments.
Against this background, the proposed regulation aims to fundamentally reconfigure the legal regime applicable to plants obtained through NGTs by introducing a binary distinction that departs from the uniform classification as GMOs established by CJEU case law. This segmentation is based on a product-oriented approach rather than on the technique used, marking a significant development in EU biosafety law.
What does the European Commission’s proposal provide?
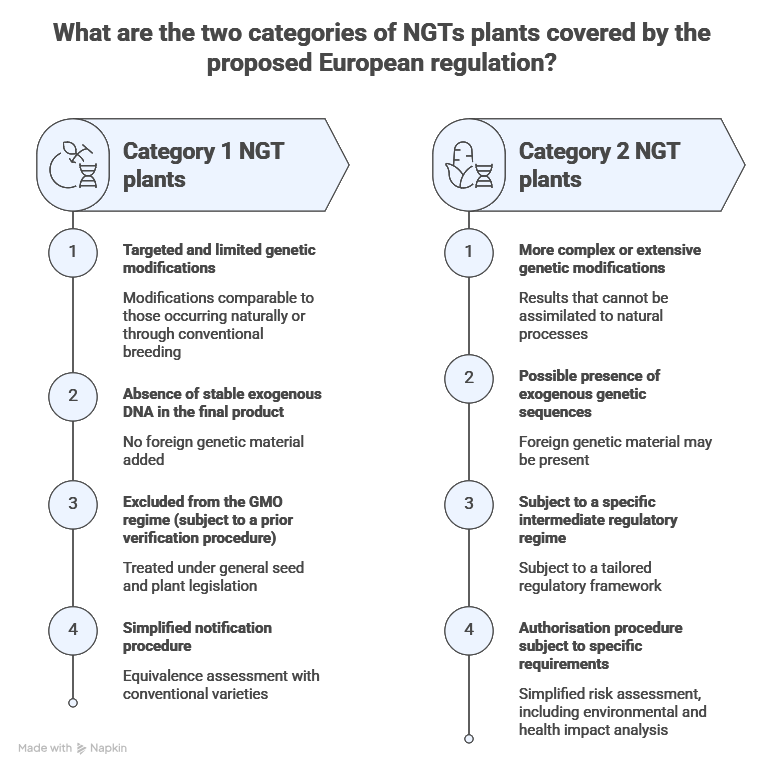
On 5 July 2023, the Commission proposed a specific regulatory framework for plants obtained by certain NGTs. The core idea is no longer to treat them uniformly as GMOs, but to distinguish between two categories based on the characteristics of the final product. The proposal thus adopts a binary structure: category 1 and category 2.
Category 1 NGT plants: towards assimilation with general seed law
Category 1 plants are characterised by targeted, limited genetic modifications that are indistinguishable from those that could occur naturally or through conventional breeding methods:
- No stable insertion of exogenous DNA into the genome
- Limited and targeted genetic changes (site-directed mutagenesis, small insertions)
- Outcomes equivalent to those achieved through conventional mutagenesis or breeding
From a legal perspective, such plants would be expressly excluded from the scope of Directive 2001/18/EC on GMOs, subject to a verification procedure. This entails several consequences such as the removal of the requirement for prior authorisation under GMO legislation, the absence of a systematic environmental assessment for GMOs, and their inclusion within the standard regime for seeds and plants.
Category 2 NGT plants: a regulated intermediate regime
Category 2 plants encompass more complex genomic modifications, particularly where:
- The modifications exceed what could occur naturally
- Exogenous genetic sequences are inserted or recombined
- The phenotypic effects involve a higher degree of uncertainty
These plants remain subject to a specific regulatory regime, although this is significantly less stringent than the standard framework for GMOs. Consequently, the risk assessment procedure is simplified and proportionate, the authorisation process is regulated but streamlined, and the traceability requirements have been adapted.
This intermediate regime reflects an intention to calibrate regulatory constraints to the level of identified risk, in line with the principle of proportionality under EU law. Nevertheless, the practical implementation of this distinction may give rise to interpretative disputes, particularly regarding classification criteria between categories 1 and 2.
A particularly sensitive aspect of the reform concerns the redefinition of traceability and consumer information requirements.
Unlike traditional GMOs, certain NGT plants, especially those in category 1, may:
- Be exempt from specific labelling requirements where equivalent to conventional varieties
- Be placed on the market within agricultural and food supply chains without formal distinction
- Not be subject to specific GMO traceability obligations
Ongoing debates
The proposed reform raises not only biosafety concerns but also broader issues relating to intellectual property, traceability, labelling, and environmental monitoring of NGT-derived plants. These issues remain particularly sensitive in light of the provisional agreement reached at EU level in December 2025 on the future NGT regulation.
Intellectual property is central to these discussions. On the one hand, plant variety rights protect new varieties while preserving breeders’ access for further innovation. On the other hand, the use of patents for certain traits obtained through NGTs raises concerns regarding market concentration and restricted access to innovation. These questions therefore extend beyond technical legal considerations and directly affect the structure of the seed market.
Traceability and labelling also remain contentious. In practice, identifying an NGT plant based solely on the final product may be difficult, complicating control mechanisms. The issue of consumer information therefore remains unresolved: some stakeholders advocate for specific labelling, while others consider such requirements unjustified for plants closely resembling conventional varieties.
Finally, post-market monitoring remains a key issue. Even where regulatory requirements are relaxed for certain NGT plants, several stakeholders advocate maintaining appropriate environmental surveillance and specific safeguards for certain agricultural sectors, particularly those seeking to exclude such techniques.
These concerns are echoed in the French political context. On 21 January 2026, a proposal for a European resolution was submitted to the French National Assembly opposing the deregulation of NGT plants and advocating for a strict European framework in terms of health, environmental, and democratic safeguards. This initiative illustrates that, despite ongoing developments at EU level, consensus remains far from achieved.
Conclusion
The proposed regulation on plants obtained through certain new genomic techniques reflects a significant shift in EU law governing plant biotechnology. By departing from the principle of systematically classifying NGTs as GMOs, it seeks to establish a more differentiated framework based on the characteristics of the final product rather than solely on the technique used. This evolution aims to enhance regulatory coherence and legal certainty for economic operators.
However, the reform does not resolve all challenges. The determination of classification criteria, the interaction with traceability and information requirements, and the implications for intellectual property, competition, and environmental monitoring remain widely debated. In this respect, the European resolution proposal submitted to the French National Assembly in January 2026 demonstrates that the choices made at EU level continue to face significant reservations, particularly in light of the precautionary principle and the protection of certain agricultural sectors.
Beyond technical considerations, the reform highlights a broader issue: the balance EU law seeks to strike between fostering innovation, safeguarding the internal market, ensuring consumer information, and protecting the environment. This balance will ultimately determine the role that NGTs will play within the European agricultural model.
Dreyfus law firm assists its clients in managing complex intellectual property cases, offering personalized advice and comprehensive operational support for the complete protection of intellectual property.
Nathalie Dreyfus with the support of the entire Dreyfus team.
Q&A
1. What are new genomic techniques (NGTs)?
NTGs refer to modern techniques that enable the targeted modification of a plant’s genome.
2. What is the difference between NGTs and GMOs?
GMOs typically involve the introduction of foreign genetic material into a plant.
NGTs, by contrast, may modify an existing gene without introducing external DNA.
3. Why did the CJEU classify NGTs under the GMO framework?
The CJEU adopted a broad interpretation of Directive 2001/18/EC, based on the precautionary principle, holding that recent genome-editing techniques lacking a history of safe use should be subject to the same regulatory framework as traditional GMOs.
4. What are the implications for agriculture?
NGTs may improve crop yields and resilience but also raise economic, legal, and societal issues.
This publication is intended to provide general guidance and highlight certain issues. It is not intended to apply to specific situations nor to constitute legal advice.