Sommaire
Introduction
Obtaining a Plant Variety Right (PVR) in the European Union from material originating in a third country is not a mere paperwork exercise. For a non-EU breeder or seed company, the principal risk is not that the variety itself proves “unfit”, but that the project fails before the PVR examination even begins, for want of regulatory anticipation.
In the majority of unsuccessful cases, the same root cause is at work: applicants begin by filing a PVR application in the belief that the rest will follow, whereas, in respect of material originating outside the EU, the filing is merely the culmination of a far broader process.
That process presupposes a number of prerequisites, such as authorization to enter the EU, the traceability of the material’s origin, and compliance with phytosanitary requirements.
This article examines, step by step, the preliminary stages preceding a PVR application where the material in question originates from countries outside the European Union.
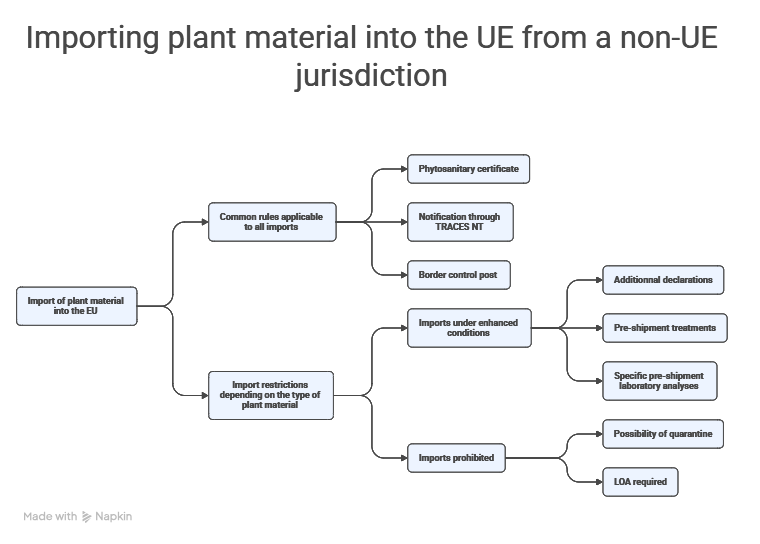
Rules governing the introduction of plant material from non-EU jurisdiction into the European territory
Before contemplating a “Plant Variety Right”, one simple question must be addressed: is it lawful to introduce the plant material into the European Union, and on what terms?
- Factors governing the entry of material into the EU
The analysis turns on several cumulative factors, derived in particular from the lists and requirements set out in Commission Implementing Regulation (EU) 2019/2072:
- Species (apple, rose, cereals, vine, etc.): not all species present the same level of risk. Certain plants are particularly susceptible to, or vectors of, regulated diseases.
- Country of origin: certain countries are deemed to present a high phytosanitary risk owing to the presence of harmful organisms therein which are absent from European territory.
- Type of material: the nature of the material plays a decisive role, since it influences the likelihood of disease transmission. Seeds, for instance, present a lower risk than a whole plant.
- Risk profile: the analysis also takes into account the harmful organisms regulated by the European Union. These include parasites, viruses, bacteria and fungi liable to cause serious damage to European crops.
When these parameters are combined, three typical situations emerge:
- Importation is authorized, subject to compliance with general rules (phytosanitary certificate, notification through TRACES NT, and clearance through a Border Control Post).
- Importation is authorized subject to enhanced conditions (additional declarations on the phytosanitary certificate, specific treatments such as disinfection, or mandatory pre-shipment analyses).
- Importation is directly prohibited.
- Common conditions applicable to any introduction of non-EU plant material into the EU
Although several import scenarios exist (authorized, conditional, direct refusal), all must satisfy certain regulatory requirements. Regulation (EU) 2016/2031 establishes the framework for protective measures against pests of plants and makes the entry of plants conditional upon:
- An official phytosanitary certificate issued by the National Plant Protection Organization (NPPO) of the country of origin (in France, the Organisation nationale de la protection des végétaux), recognized in accordance with international standards (IPPC), certifying that the plant material complies with EU phytosanitary requirements.
- A notification through the European TRACES NT platform, which enables sensitive consignments such as plants and plant materials to be notified and traced.
- Mandatory clearance through a Border Control Post (BCP).
- The phytosanitary certificate
Securing a phytosanitary certificate is no mere administrative formality: it presupposes compliance with stringent upstream conditions, controlled by the competent authority of the country of origin.
In practice, several cumulative requirements must be met:
- Official inspection of the material: the plant material must be inspected by the National Plant Protection Organization in order to verify the absence of regulated harmful organisms.
- Traceability of origin: the origin of the material must be clearly identified and documented (production site, growing conditions, treatment history).
- Compliance with EU-specific requirements: certain species or origins call for additional declarations on the certificate (for example, freedom from a specified pest, or compliance with particular cultivation conditions).
- Performance of pre-shipment tests or treatments: depending on the case, laboratory analyses, phytosanitary treatments or specific procedures may be required prior to export.
- Documentary consistency: the information appearing on the certificate (species, quantity, origin, type of material) must correspond strictly to the consignment shipped.
In practice, the phytosanitary certificate is therefore the outcome of an upstream control process, and not a mere declaratory document.
Any non-conformity — whether an incomplete certificate, a missing entry, or an inconsistency between the documents and the material itself — may give rise to immediate consequences upon entry into EU territory, including refusal of entry, detention or quarantine, or even the destruction of the material in accordance with the provisions of Regulation (EU) 2017/625.
- The customs dimension
The introduction of plant material into the European Union is governed by the general regulatory framework laid down in the Union Customs Code (Regulation (EU) No 952/2013). In practical terms, the operator must:
- Lodge a customs declaration in compliance with the Union Customs Code, selecting the appropriate procedure (release for free circulation, temporary admission, etc.) according to the project (trials, quarantine, subsequent commercial exploitation);
- Determine the appropriate tariff code (TARIC) for the type of material concerned (seeds, plants, in vitro cultures, etc.);
- Calculate the customs duties and import VAT.
In practice, the phytosanitary and customs aspects operate as two parallel filters: a consignment may be compliant on the phytosanitary front but blocked at customs (and vice versa). For a non-EU operator, it is therefore essential to prepare a coherent file in which the phytosanitary and customs information dovetail without contradiction.
The case of imports into the EU subject to enhanced conditions
In this scenario, the material remains admissible into the territory of the Union, but its introduction is subject to compliance with specific requirements set out in Annex VII of Commission Implementing Regulation (EU) 2019/2072, which vary according to the species, the origin and the phytosanitary risk profile. These requirements fall, principally, into three categories.
They take the form, in the first place, of additional declarations to be entered on the phytosanitary certificate, by which the NPPO of the country of origin attests that a specific phytosanitary condition is satisfied. By way of illustration, plants for planting figuring among the hosts of Xylella fastidiosa may enter the Union only where the certificate attests, in accordance with the options provided for, that the area or the production site is recognized as free from the organism, that the site is physically protected against the introduction of vectors, or that the plants have been subjected there to regular official inspections coupled with analyses.
They may also take the form of pre-shipment treatments. In such cases, admissibility is conditional upon the application, prior to dispatch, of technical measures laid down by the regulation, such as cold treatment under a specified temperature and duration regime for fruits of Citrus sinensis Pers. The treatment applied must be expressly stated on the phytosanitary certificate, in the section reserved for disinfestation or disinfection.
They may, lastly, consist of pre-shipment laboratory analyses, where Annex VII requires official sampling and diagnosis prior to dispatch in order to confirm the absence of a specified organism in the lot concerned. The results must be contemporaneous with the consignment and traceable to the lot dispatched, failing which documentary non-conformity will arise.
These requirements are implemented upstream, in the country of origin, under the responsibility of the NPPO, and are verified upon clearance through the Border Control Post on the basis of the phytosanitary certificate together with documentary, identity and physical checks.
The hypothesis of an importation prohibited within the territory of the European Union
Where direct importation is impossible, quarantine may become the gateway to the European Union.
- What is plant quarantine?
Quarantine is the holding of plant material in an approved facility, under the supervision of the competent authorities, in order to verify the absence of regulated pests and diseases, where the material is introduced for scientific, trial or breeding purposes, prior to any subsequent movement within the European Union.
In practice, the process unfolds in several stages:
- Prior reservation of a place at a quarantine station.
- Reception of the material and, where applicable, performance of technical operations (grafting, multiplication, etc.).
- A series of phytosanitary analyses and inspections.
- Isolation and surveillance for a period generally ranging between one and two years.
- Release from quarantine, where everything is compliant, allowing transfer of the material to a DUS (Distinctness, Uniformity, Stability) trial site within the EU.
- The need for an LOA.
In schemes involving quarantine in France, the central instrument is, in most cases, the Letter of Authorization (LOA) issued by the competent authority. It is an official document evidencing the agreement of the phytosanitary authorities to the introduction, within a strictly controlled framework, of plant material regarded as sensitive.
Securing such an official document presupposes:
- The reservation of quarantine places at the approved station.
- The compilation of a technical file detailing the species, type of material, origin, known phytosanitary status, intended use, and so forth.
- Discussions with the phytosanitary authorities to ensure that the conditions of containment and monitoring are deemed sufficient.
The LOA authorizes the importation of the material to the designated quarantine station and governs the conditions of its subsequent movement within the EU once the controls have been lifted (notably to DUS trial sites). Commission Delegated Regulation (EU) 2019/829 specifies the conditions for the derogation and for movements of plants for scientific, trial or breeding purposes provided for in Regulation (EU) 2016/2031, and provides the legal basis for obtaining such a Letter of Authorisation.
It bears emphasizing, however, that the LOA is not to be conflated with an import authorization in the strict sense: it constitutes a circumscribed derogation enabling introduction into quarantine, without prejudice to compliance with the other applicable regulatory requirements, in particular as regards phytosanitary certification and controls at points of entry.
- Practical implications for the PVR
Quarantine is a key regulatory step governing the introduction of plant material, principally for scientific, trial or breeding purposes, and not for direct placing on the market in the European Union. It is intended to prevent the introduction of harmful organisms and to verify the phytosanitary status of the material before any subsequent movement.
- It may, in certain cases, allow the introduction of material that would otherwise be prohibited from direct importation.
- It significantly extends timelines, often by at least one growing cycle.
- It may call for additional material (rootstocks, controls), which is itself subject to the phytosanitary requirements.
- It carries an element of uncertainty, since the material may be refused if regulated organisms are detected.
Within a PVR project involving material of non-EU origin, integrating quarantine into the planning phase from the outset is decisive: this is, in many cases, what makes the difference between a theoretical filing and a right that is genuinely capable of exploitation.
Conclusion
Securing plant variety protection in Europe from material originating outside the EU is, first and foremost, not a question of paperwork but of regulatory preparation. Anticipating territorial access, securing the lawfulness of the origin, and organizing robust traceability now lie at the heart of the strategy underlying any PVR application.
A successful project is one in which the timetable of DUS trials is aligned with these constraints, and in which each stage of the introduction of the material can be evidenced. On that condition, the certificate obtained will not merely be valid, but truly enforceable in practice.
Dreyfus law firm assists clients in the management of complex intellectual property matters, providing tailored advice and comprehensive operational support for the integral protection of intellectual property rights.
Nathalie Dreyfus with the support of the entire Dreyfus team.
FAQ
1. Can DUS trials be undertaken before quarantine is lifted?
So long as quarantine has not been lifted, the material remains confined and is legally unavailable for the trials.
2. Can an error in the LOA stall a project?
Yes, an inconsistency (species, quantity, origin, intended use) may result in refusal of entry, detention of the material, or even its destruction.
3. Does the LOA cover the technical operations carried out in quarantine (grafting, multiplication)?
Not directly: it authorizes the introduction, but the technical operations must be set out in the protocol approved with the station and the authorities.
4. Can a PVR project be secured before the material to be introduced has been precisely identified?
Only with great difficulty. The requirements (phytosanitary, LOA, quarantine) depend heavily on the precise type of material, which makes any approximate planning hazardous.
This publication is intended to provide general guidance to the public and to highlight certain issues. It is not intended to apply to particular situations and does not constitute legal advice.